Would you care for a pragmatic understanding of cost-effectiveness for new #cardiovascular drugs? https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen">
In our new @Heart_BMJ article, we use 3 case studies to clarify key concepts like cost-effectiveness & affordability: #tafamidis, #PCSK9i & #DOAC.
 https://abs.twimg.com/emoji/v2/... draggable="false" alt="🧵" title="Thread" aria-label="Emoji: Thread">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🧵" title="Thread" aria-label="Emoji: Thread">
https://tinyurl.com/BMJcea ">https://tinyurl.com/BMJcea&qu...
In our new @Heart_BMJ article, we use 3 case studies to clarify key concepts like cost-effectiveness & affordability: #tafamidis, #PCSK9i & #DOAC.
https://tinyurl.com/BMJcea ">https://tinyurl.com/BMJcea&qu...
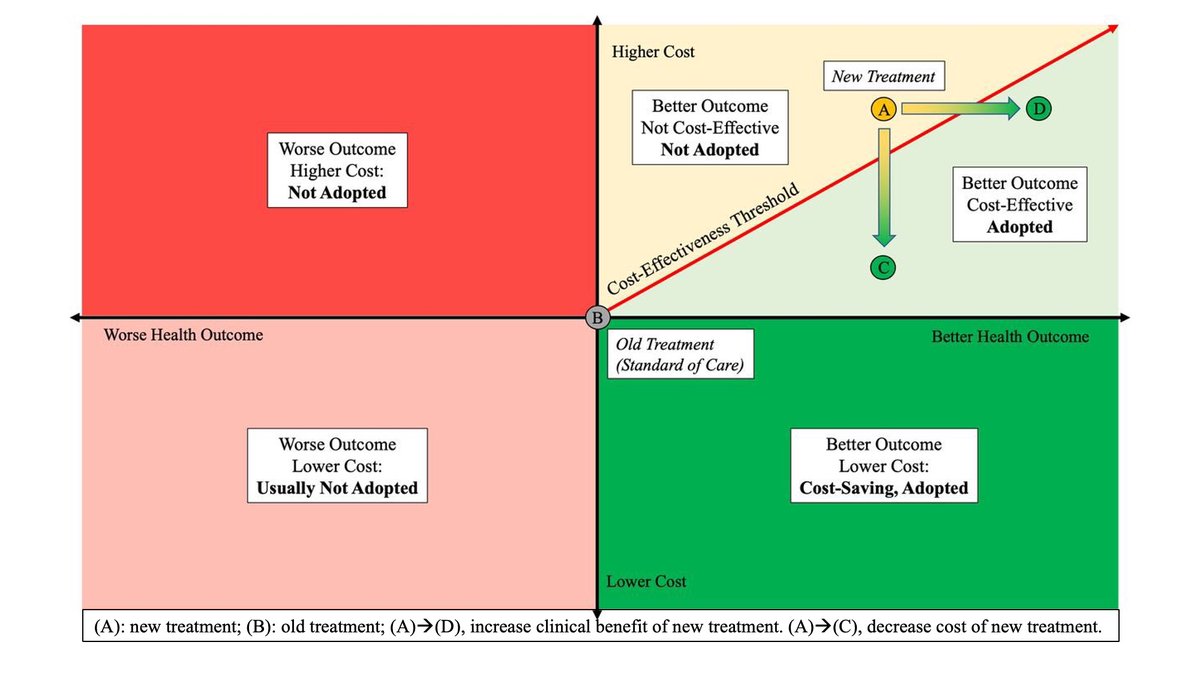
2) How do we decide if a new therapy represents societal value for money? Well, it depends on how we define “value”.
3) We often use #ICER. The Incremental Cost Effectiveness Ratio evaluates the Cost (numerator) and "Benefit" (denominator) of new drug  https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht"> compared to old drug
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht"> compared to old drug  https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht">. If #ICER is lower than what we are willing to pay as a society (in the US ~ $100K/QALY),the new
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht">. If #ICER is lower than what we are willing to pay as a society (in the US ~ $100K/QALY),the new  https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> is cost-effective.
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> is cost-effective.
4)Did you say #QALY? Quality-Adjusted Life-Years can compare "apples to oranges", i.e. standardize the benefit of different clinical outcomes (ex. avoiding a stroke vs a bleed). It includes prolonged survival & quality of life on a utility scale from 0 (death) to 1 (full health).
5) Next, let’s define cost. Key caveat: Cost = TOTAL Cost = cost of new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> PLUS all downstream expenses: monitoring, side effects, additional healthcare due to
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> PLUS all downstream expenses: monitoring, side effects, additional healthcare due to  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben"> survival, but also savings when new
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben"> survival, but also savings when new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten">cardiovascular events. Most new
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten">cardiovascular events. Most new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> likely
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> likely  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">net costs. Is it worth the benefit?
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">net costs. Is it worth the benefit?
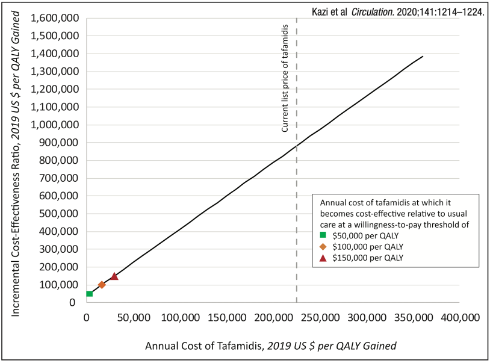
7)In 2019, #Tafamidis entered market at $225,000/yr  https://abs.twimg.com/emoji/v2/... draggable="false" alt="😱" title="Vor Angst schreiendes Gesicht" aria-label="Emoji: Vor Angst schreiendes Gesicht">, but offered impressive 1.3 QALY gain over lifetime for #amyloid pts (compared w/ no other available rx).This generates #ICER $880,000/QALY vs usual care. To meet $100,000/QALY, cost would need to
https://abs.twimg.com/emoji/v2/... draggable="false" alt="😱" title="Vor Angst schreiendes Gesicht" aria-label="Emoji: Vor Angst schreiendes Gesicht">, but offered impressive 1.3 QALY gain over lifetime for #amyloid pts (compared w/ no other available rx).This generates #ICER $880,000/QALY vs usual care. To meet $100,000/QALY, cost would need to  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> to $16,650/yr (93% drop).
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> to $16,650/yr (93% drop).
8) In 2016, #PCSK9i entered market at $14,500/yr  https://abs.twimg.com/emoji/v2/... draggable="false" alt="😬" title="Grimasse schneidendes Gesicht" aria-label="Emoji: Grimasse schneidendes Gesicht"> -> #ICER $415,000/QALY (vs statin+Zetia)
https://abs.twimg.com/emoji/v2/... draggable="false" alt="😬" title="Grimasse schneidendes Gesicht" aria-label="Emoji: Grimasse schneidendes Gesicht"> -> #ICER $415,000/QALY (vs statin+Zetia)  https://abs.twimg.com/emoji/v2/... draggable="false" alt="👎" title="Daumen runter" aria-label="Emoji: Daumen runter">. Thanks to market pressure, in 2018 both manufacturers
https://abs.twimg.com/emoji/v2/... draggable="false" alt="👎" title="Daumen runter" aria-label="Emoji: Daumen runter">. Thanks to market pressure, in 2018 both manufacturers  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> price by 60% to $4,500/yr -> #ICER $56,600/QALY for pts w/ high #CV risk = Cost-effective
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> price by 60% to $4,500/yr -> #ICER $56,600/QALY for pts w/ high #CV risk = Cost-effective  https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. BUT are these meds affordable?
https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. BUT are these meds affordable?
9)Affordable to who? For patients, affordability = out of pocket https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen">. For payors=budget impact, i.e. net change in expenditure if all eligible pts receive new
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen">. For payors=budget impact, i.e. net change in expenditure if all eligible pts receive new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">. So even for cost-effective
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">. So even for cost-effective https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">, rare vs common disease matters! Often,
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">, rare vs common disease matters! Often,  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost for payors =
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost for payors = https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">co-pay = unaffordable for pts.
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">co-pay = unaffordable for pts.
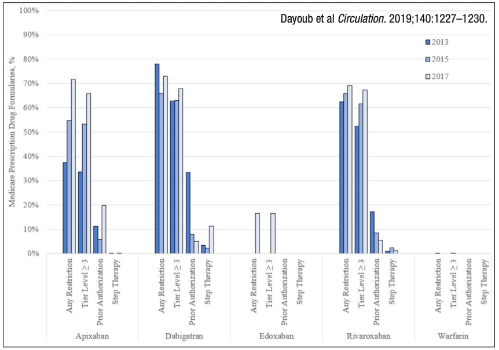
10)Total spending matters too, as in #DOAC vs #warfarin. #ICER for DOAC vs warfarin: $54,000/QALY https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. But as DOAC adoption
https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. But as DOAC adoption https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">, Medicare spending on anticoagulation also
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">, Medicare spending on anticoagulation also https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">from $750Million in 2012 to $4.7Billion in 2016. Unsurprisingly, Medicare formularies
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">from $750Million in 2012 to $4.7Billion in 2016. Unsurprisingly, Medicare formularies https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost-sharing/prior auths.
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost-sharing/prior auths.
11) Thus, #DOAC represent a perfect example of a  https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> that is both clinically effective AND cost-effective, but may nonetheless be unaffordable for many patients. Certainly, upcoming patent expiration in 2022 will likely
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> that is both clinically effective AND cost-effective, but may nonetheless be unaffordable for many patients. Certainly, upcoming patent expiration in 2022 will likely  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> budget impact,
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> budget impact,  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> out of pocket
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> out of pocket  https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen">, and
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen">, and  https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben"> affordability.
https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben"> affordability.
12)Take-home #1: clinicians should complement rigorous understanding of clinical trial results w/ cost-effectiveness principles, to understand both health needs & economic priorities of pts & health systems. This ensures high-cost new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> reach pts most likely to benefit from them.
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> reach pts most likely to benefit from them.
13) Take-home #2: the 60% historical price reduction of #PCSK9i exemplifies how rigorous cost-effectiveness analyses can help decrease cost, promote access, and mitigate healthcare disparities – especially if timely conducted to match trial release and guide pricing negotiations.
14)Thank you @kardiologykazi and Blake Liu for sharing your wisdom & expertise on how to interpret cost-effectiveness analyses and their broader societal implications on affordability. Thank you also to @SmithBIDMC for the support on this project! #MedTwitter #CardioTwitter

 Read on Twitter
Read on Twitter https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen"> In our new @Heart_BMJ article, we use 3 case studies to clarify key concepts like cost-effectiveness & affordability: #tafamidis, #PCSK9i & #DOAC.https://abs.twimg.com/emoji/v2/... draggable="false" alt="🧵" title="Thread" aria-label="Emoji: Thread"> https://tinyurl.com/BMJcea&qu..." title="Would you care for a pragmatic understanding of cost-effectiveness for new #cardiovascular drugs?https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen"> In our new @Heart_BMJ article, we use 3 case studies to clarify key concepts like cost-effectiveness & affordability: #tafamidis, #PCSK9i & #DOAC.https://abs.twimg.com/emoji/v2/... draggable="false" alt="🧵" title="Thread" aria-label="Emoji: Thread"> https://tinyurl.com/BMJcea&qu..." class="img-responsive" style="max-width:100%;"/>
https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen"> In our new @Heart_BMJ article, we use 3 case studies to clarify key concepts like cost-effectiveness & affordability: #tafamidis, #PCSK9i & #DOAC.https://abs.twimg.com/emoji/v2/... draggable="false" alt="🧵" title="Thread" aria-label="Emoji: Thread"> https://tinyurl.com/BMJcea&qu..." title="Would you care for a pragmatic understanding of cost-effectiveness for new #cardiovascular drugs?https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette">https://abs.twimg.com/emoji/v2/... draggable="false" alt="💵" title="Banknote mit Dollar-Zeichen" aria-label="Emoji: Banknote mit Dollar-Zeichen"> In our new @Heart_BMJ article, we use 3 case studies to clarify key concepts like cost-effectiveness & affordability: #tafamidis, #PCSK9i & #DOAC.https://abs.twimg.com/emoji/v2/... draggable="false" alt="🧵" title="Thread" aria-label="Emoji: Thread"> https://tinyurl.com/BMJcea&qu..." class="img-responsive" style="max-width:100%;"/>
 compared to old drug https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht">. If #ICER is lower than what we are willing to pay as a society (in the US ~ $100K/QALY),the new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> is cost-effective." title="3) We often use #ICER. The Incremental Cost Effectiveness Ratio evaluates the Cost (numerator) and "Benefit" (denominator) of new drug https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht"> compared to old drug https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht">. If #ICER is lower than what we are willing to pay as a society (in the US ~ $100K/QALY),the new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> is cost-effective." class="img-responsive" style="max-width:100%;"/>
compared to old drug https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht">. If #ICER is lower than what we are willing to pay as a society (in the US ~ $100K/QALY),the new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> is cost-effective." title="3) We often use #ICER. The Incremental Cost Effectiveness Ratio evaluates the Cost (numerator) and "Benefit" (denominator) of new drug https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht"> compared to old drug https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚨" title="Polizeiautos mit drehendem Licht" aria-label="Emoji: Polizeiautos mit drehendem Licht">. If #ICER is lower than what we are willing to pay as a society (in the US ~ $100K/QALY),the new https://abs.twimg.com/emoji/v2/... draggable="false" alt="💊" title="Tablette" aria-label="Emoji: Tablette"> is cost-effective." class="img-responsive" style="max-width:100%;"/>
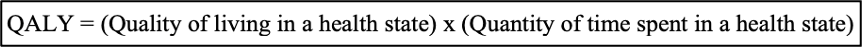
 , but offered impressive 1.3 QALY gain over lifetime for #amyloid pts (compared w/ no other available rx).This generates #ICER $880,000/QALY vs usual care. To meet $100,000/QALY, cost would need to https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> to $16,650/yr (93% drop)." title="7)In 2019, #Tafamidis entered market at $225,000/yr https://abs.twimg.com/emoji/v2/... draggable="false" alt="😱" title="Vor Angst schreiendes Gesicht" aria-label="Emoji: Vor Angst schreiendes Gesicht">, but offered impressive 1.3 QALY gain over lifetime for #amyloid pts (compared w/ no other available rx).This generates #ICER $880,000/QALY vs usual care. To meet $100,000/QALY, cost would need to https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> to $16,650/yr (93% drop)." class="img-responsive" style="max-width:100%;"/>
, but offered impressive 1.3 QALY gain over lifetime for #amyloid pts (compared w/ no other available rx).This generates #ICER $880,000/QALY vs usual care. To meet $100,000/QALY, cost would need to https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> to $16,650/yr (93% drop)." title="7)In 2019, #Tafamidis entered market at $225,000/yr https://abs.twimg.com/emoji/v2/... draggable="false" alt="😱" title="Vor Angst schreiendes Gesicht" aria-label="Emoji: Vor Angst schreiendes Gesicht">, but offered impressive 1.3 QALY gain over lifetime for #amyloid pts (compared w/ no other available rx).This generates #ICER $880,000/QALY vs usual care. To meet $100,000/QALY, cost would need to https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> to $16,650/yr (93% drop)." class="img-responsive" style="max-width:100%;"/>
 -> #ICER $415,000/QALY (vs statin+Zetia) https://abs.twimg.com/emoji/v2/... draggable="false" alt="👎" title="Daumen runter" aria-label="Emoji: Daumen runter">. Thanks to market pressure, in 2018 both manufacturers https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> price by 60% to $4,500/yr -> #ICER $56,600/QALY for pts w/ high #CV risk = Cost-effective https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. BUT are these meds affordable?" title="8) In 2016, #PCSK9i entered market at $14,500/yr https://abs.twimg.com/emoji/v2/... draggable="false" alt="😬" title="Grimasse schneidendes Gesicht" aria-label="Emoji: Grimasse schneidendes Gesicht"> -> #ICER $415,000/QALY (vs statin+Zetia) https://abs.twimg.com/emoji/v2/... draggable="false" alt="👎" title="Daumen runter" aria-label="Emoji: Daumen runter">. Thanks to market pressure, in 2018 both manufacturers https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> price by 60% to $4,500/yr -> #ICER $56,600/QALY for pts w/ high #CV risk = Cost-effective https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. BUT are these meds affordable?" class="img-responsive" style="max-width:100%;"/>
-> #ICER $415,000/QALY (vs statin+Zetia) https://abs.twimg.com/emoji/v2/... draggable="false" alt="👎" title="Daumen runter" aria-label="Emoji: Daumen runter">. Thanks to market pressure, in 2018 both manufacturers https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> price by 60% to $4,500/yr -> #ICER $56,600/QALY for pts w/ high #CV risk = Cost-effective https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. BUT are these meds affordable?" title="8) In 2016, #PCSK9i entered market at $14,500/yr https://abs.twimg.com/emoji/v2/... draggable="false" alt="😬" title="Grimasse schneidendes Gesicht" aria-label="Emoji: Grimasse schneidendes Gesicht"> -> #ICER $415,000/QALY (vs statin+Zetia) https://abs.twimg.com/emoji/v2/... draggable="false" alt="👎" title="Daumen runter" aria-label="Emoji: Daumen runter">. Thanks to market pressure, in 2018 both manufacturers https://abs.twimg.com/emoji/v2/... draggable="false" alt="⬇️" title="Pfeil nach unten" aria-label="Emoji: Pfeil nach unten"> price by 60% to $4,500/yr -> #ICER $56,600/QALY for pts w/ high #CV risk = Cost-effective https://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. BUT are these meds affordable?" class="img-responsive" style="max-width:100%;"/>
 . But as DOAC adoptionhttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">, Medicare spending on anticoagulation alsohttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">from $750Million in 2012 to $4.7Billion in 2016. Unsurprisingly, Medicare formularieshttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost-sharing/prior auths." title="10)Total spending matters too, as in #DOAC vs #warfarin. #ICER for DOAC vs warfarin: $54,000/QALYhttps://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. But as DOAC adoptionhttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">, Medicare spending on anticoagulation alsohttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">from $750Million in 2012 to $4.7Billion in 2016. Unsurprisingly, Medicare formularieshttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost-sharing/prior auths." class="img-responsive" style="max-width:100%;"/>
. But as DOAC adoptionhttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">, Medicare spending on anticoagulation alsohttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">from $750Million in 2012 to $4.7Billion in 2016. Unsurprisingly, Medicare formularieshttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost-sharing/prior auths." title="10)Total spending matters too, as in #DOAC vs #warfarin. #ICER for DOAC vs warfarin: $54,000/QALYhttps://abs.twimg.com/emoji/v2/... draggable="false" alt="👍" title="Daumen hoch" aria-label="Emoji: Daumen hoch">. But as DOAC adoptionhttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">, Medicare spending on anticoagulation alsohttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">from $750Million in 2012 to $4.7Billion in 2016. Unsurprisingly, Medicare formularieshttps://abs.twimg.com/emoji/v2/... draggable="false" alt="⬆️" title="Pfeil nach oben" aria-label="Emoji: Pfeil nach oben">cost-sharing/prior auths." class="img-responsive" style="max-width:100%;"/>