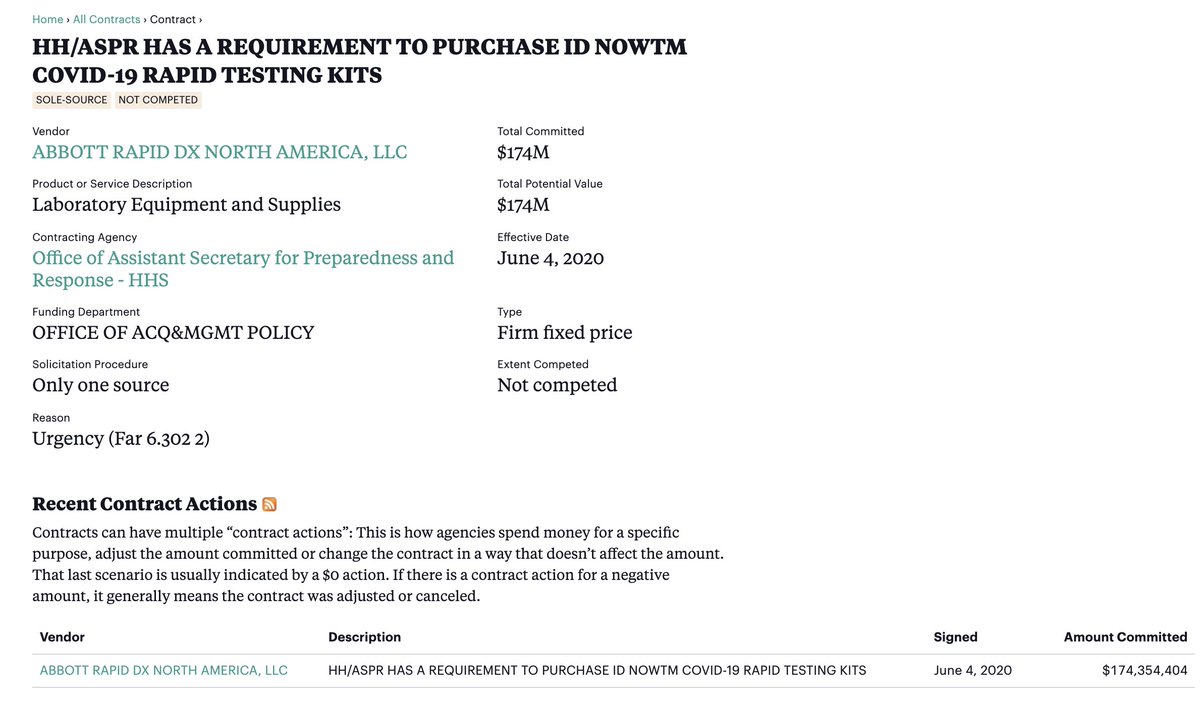
One might begin by asking HHS ASPR, Robert Kadlec, to explain the rationale behind their "requirement to purchase” $174M worth of Abbott ID NOW testing kits.
A follow-up question might be: “Were you aware of the research study showing that these test kits missed nearly half of positive cases?” https://www.cnbc.com/2020/05/13/abbotts-rapid-coronavirus-test-misses-positive-cases-raising-questions-nyu-study-finds.html">https://www.cnbc.com/2020/05/1...
It would be worth pressing the issue by asking: Why issue a ’requirement’ to purchase *knowingly* faulty tests 3 weeks *after* the FDA issued a public warning citing multiple reports of problems with this testing platform? https://www.medscape.com/viewarticle/930599">https://www.medscape.com/viewartic...
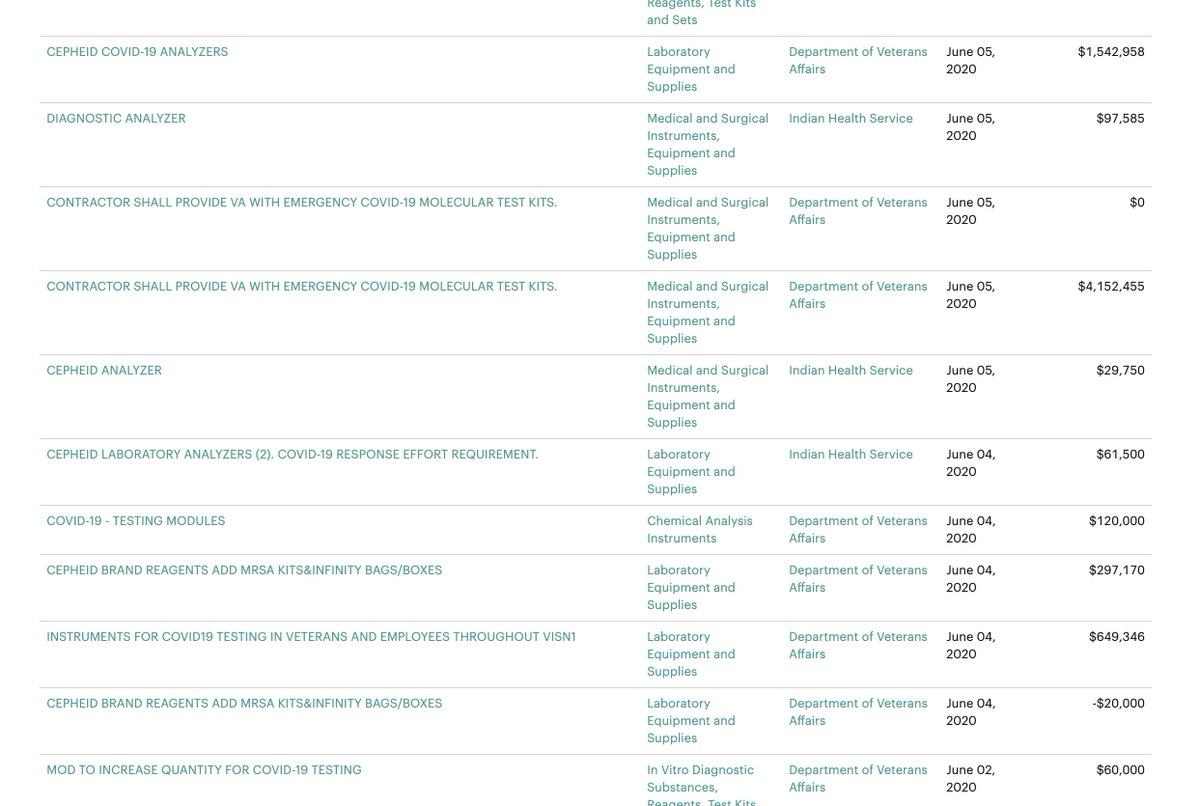
Why did no such “requirement” exist for tests made by Abbott’s primary competitor despite the fact that both the VA and parts of the Indian Health Service were making their preference clear through repeat orders— before and after the $174M contract.
see: https://projects.propublica.org/coronavirus-contracts/vendors/cepheid">https://projects.propublica.org/coronavir...
see: https://projects.propublica.org/coronavirus-contracts/vendors/cepheid">https://projects.propublica.org/coronavir...
More to the point: Why was there such as disparity in the Trump Administration’s financial commitment to a faulty testing platform despite multiple public reports and an FDA notice?
sources:
https://projects.propublica.org/coronavirus-contracts/vendors/cepheid
https://projects.propublica.org/coronavir... href=" https://projects.propublica.org/coronavirus-contracts/vendors/abbott-rapid-dx-north-america-llc">https://projects.propublica.org/coronavir...
sources:
https://projects.propublica.org/coronavirus-contracts/vendors/cepheid
Furthermore: why did HHS deem it appropriate to effectively circumvent the FDA by instructing IHS continue using the Abbott ID NOW system as-is?
https://indiancountrytoday.com/news/fda-warns-of-test-inaccuracies-ihs-finds-acceptable-r-44VHfUGkG8NFYVVwhgFw">https://indiancountrytoday.com/news/fda-...
https://indiancountrytoday.com/news/fda-warns-of-test-inaccuracies-ihs-finds-acceptable-r-44VHfUGkG8NFYVVwhgFw">https://indiancountrytoday.com/news/fda-...
Why were medical clinics serving Native communities boxed into going along with HHS (via IHS) vs. siding with the FDA, and experts in the public and private sectors? https://bringmethenews.com/minnesota-news/fond-du-lac-band-stops-using-covid-19-test-over-accuracy-concerns">https://bringmethenews.com/minnesota...
Over at GSA: @GSAEmily should explain how this contract does NOT violate FAR 6.302-2, which places clear limits on the use of non-competitive contracts. At least two such limitations appear to be violated in this case.
Src: #FAR_6_302_2">https://www.acquisition.gov/far/part-6 #FAR_6_302_2">https://www.acquisition.gov/far/part-...
Src: #FAR_6_302_2">https://www.acquisition.gov/far/part-6 #FAR_6_302_2">https://www.acquisition.gov/far/part-...
Emily should be able to answer: “Why haven’t the justifications for non-competitive purchasing been produced and published as required under FAR 6.302?”
see: #FAR_6_302_2">https://www.acquisition.gov/far/part-6 #FAR_6_302_2">https://www.acquisition.gov/far/part-...
see: #FAR_6_302_2">https://www.acquisition.gov/far/part-6 #FAR_6_302_2">https://www.acquisition.gov/far/part-...
“As a minimum" Emily should be able to provide the “estimated cost, or other rationale as to the extent and nature of the harm to the Government” and “determination that the use of a sole-source contract is in the best interest of the agency concerned”
see #FAR_6_303_2">https://www.acquisition.gov/far/part-6 #FAR_6_303_2">https://www.acquisition.gov/far/part-...
see #FAR_6_303_2">https://www.acquisition.gov/far/part-6 #FAR_6_303_2">https://www.acquisition.gov/far/part-...
A justification for that $174M not-competed contract is nowhere on HHS, http://SAM.gov"> http://SAM.gov , and http://FPDS.gov"> http://FPDS.gov — a fact that in and of itself is in violation of multiple regulations.
It would be worth asking Emily about this, too.
see: #FAR_6_305">https://www.acquisition.gov/far/part-6 #FAR_6_305">https://www.acquisition.gov/far/part-...
It would be worth asking Emily about this, too.
see: #FAR_6_305">https://www.acquisition.gov/far/part-6 #FAR_6_305">https://www.acquisition.gov/far/part-...
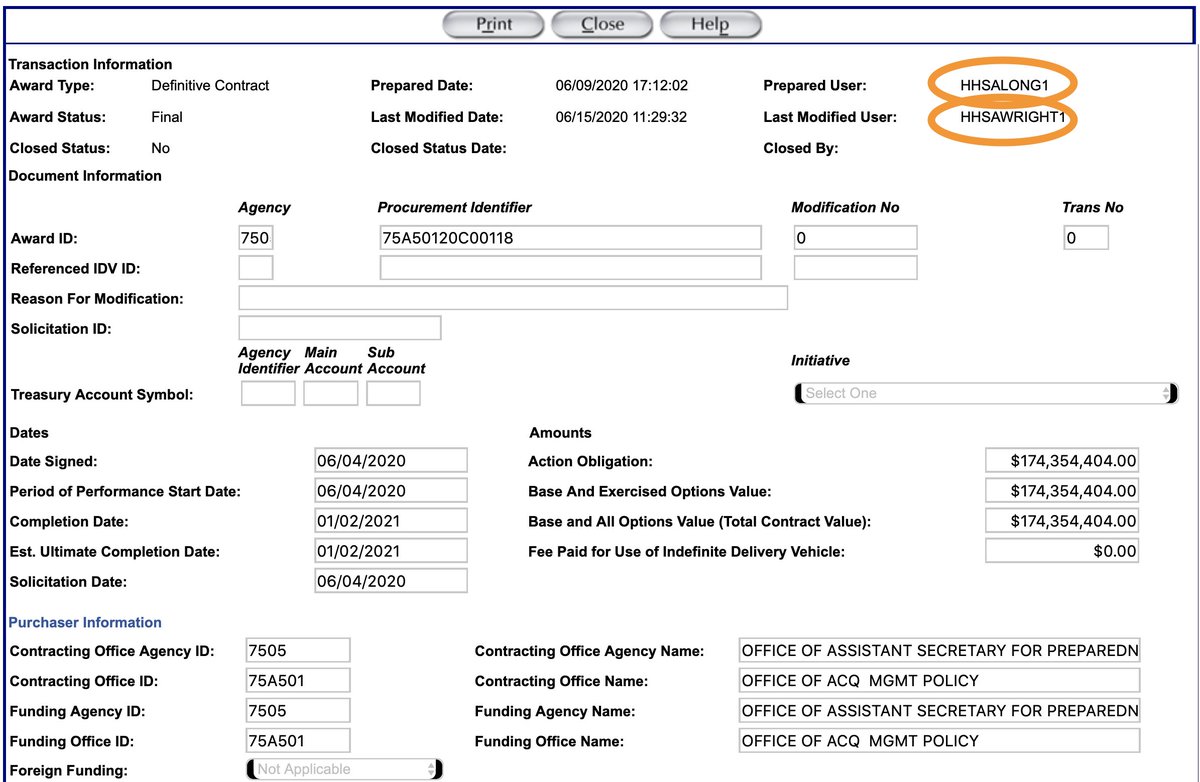
If Emily doesn’t have access to the written justifications, there are two individuals over at HHS that can point investigative journalists in the right direction (see image)
While chatting with “along” and/or “awright” at HHS, it would be important to ask them *who* authorized the contract, and to confirm that this person had authority to do so.
After all, that authority cannot be delegated under federal law. https://abs.twimg.com/emoji/v2/... draggable="false" alt="🤔" title="Denkendes Gesicht" aria-label="Emoji: Denkendes Gesicht">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🤔" title="Denkendes Gesicht" aria-label="Emoji: Denkendes Gesicht">
#FAR_6_304">https://www.acquisition.gov/far/part-6 #FAR_6_304">https://www.acquisition.gov/far/part-...
After all, that authority cannot be delegated under federal law.
#FAR_6_304">https://www.acquisition.gov/far/part-6 #FAR_6_304">https://www.acquisition.gov/far/part-...
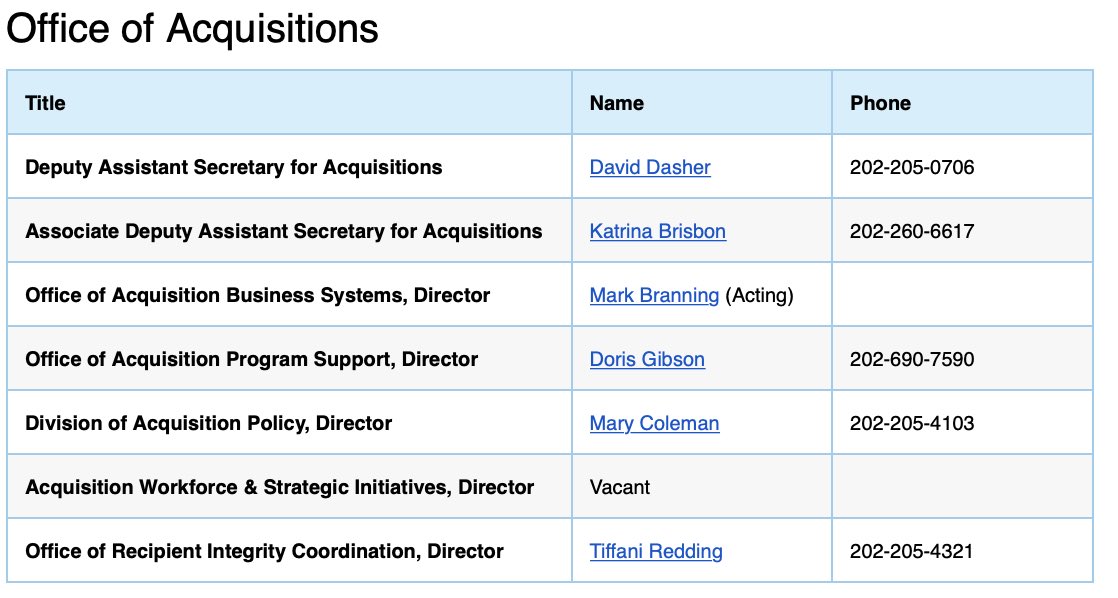
The only person who could legally authorize this contract is *Acting* Assistant Secretary for Financial Resources (ASFR) and *Acting* Chief Financial Officer for HHS, Jennifer Moughalian.
see:
https://www.hhs.gov/about/leadership/jennifer-moughalian/index.html
https://www.hhs.gov/about/lea... href=" https://www.hhs.gov/about/agencies/asfr/key-personnel/index.html">https://www.hhs.gov/about/age...
see:
https://www.hhs.gov/about/leadership/jennifer-moughalian/index.html
While chatting with HHS ASFR/CFO Moughalian, investigative journalists would do well to ask about this other thread that describes a deal worth $100M for #COVID19 testing sites for Iowa
For the same reasons, that contract would need approval at the highest levels of HHS, as well https://twitter.com/datadrivenmd/status/1326882821877624834">https://twitter.com/datadrive...
For the same reasons, that contract would need approval at the highest levels of HHS, as well https://twitter.com/datadrivenmd/status/1326882821877624834">https://twitter.com/datadrive...
Clarification: HHS ASPR Robert Kadlec may also be able to authorize the contract under the law. So, if journalists can’t reach Moughalian or Kadlec, they should try connecting w/ David M. Kelly (public records link him to #COVID19 purchases for ASPR)
see: https://govtribe.com/file/government-file/grainger-75a50120p00040-hood-sleeve-grainger-signed-3-18-20-dot-pdf">https://govtribe.com/file/gove...
see: https://govtribe.com/file/government-file/grainger-75a50120p00040-hood-sleeve-grainger-signed-3-18-20-dot-pdf">https://govtribe.com/file/gove...
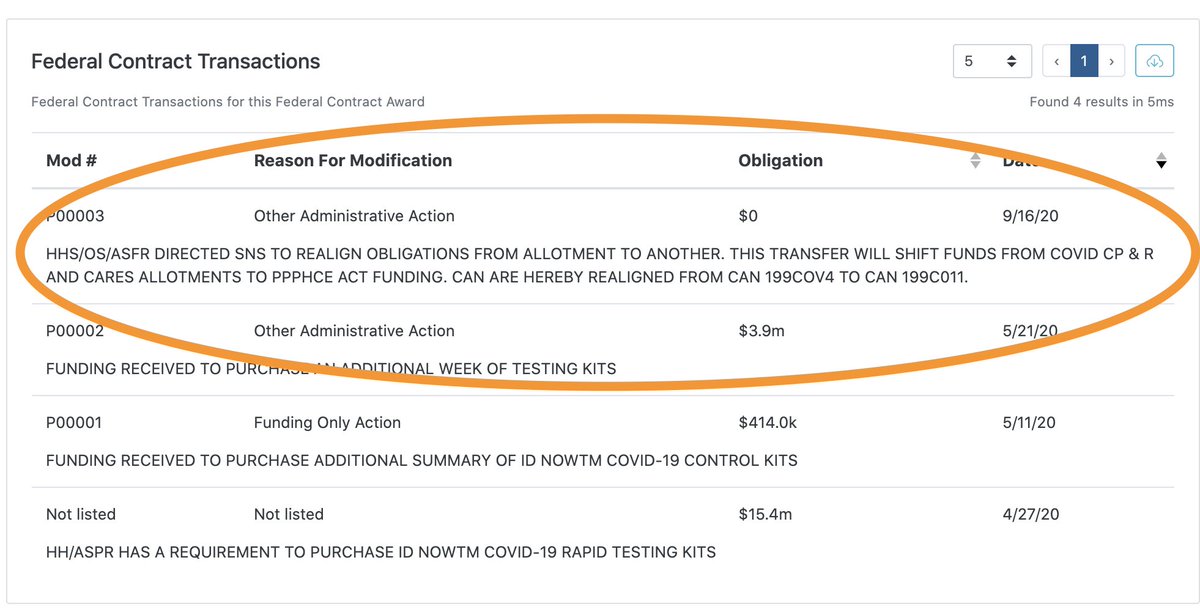
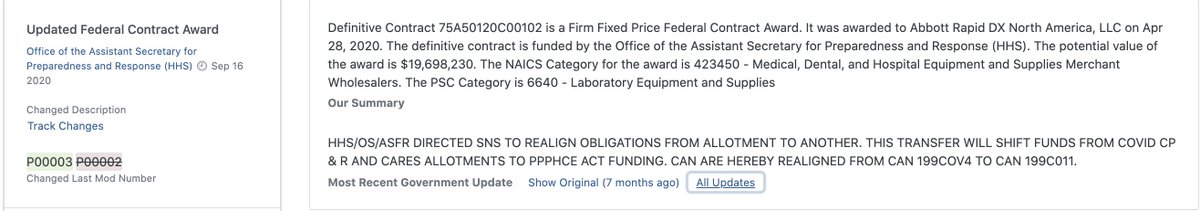
Last (for now) but not least, there’s this peculiar description in contract ID 75A50120C00102, which suggests some level of coordination between senior HHS’s chief financial officer and the Treasury…
see: https://www.usaspending.gov/award/CONT_AWD_75A50120C00102_7505_-NONE-_-NONE-">https://www.usaspending.gov/award/CON...
see: https://www.usaspending.gov/award/CONT_AWD_75A50120C00102_7505_-NONE-_-NONE-">https://www.usaspending.gov/award/CON...
…This is the only transaction with this annotation that I could find, but there are other sole-source, non-competed contracts that have similar “funding-only” transactions in the 6-8 weeks…
In light of Treasury Sec. Mnuchin’s move to shift money around #COVID19 relief programs, it’s worth asking Mnuchin why the sudden rush? https://www.cnbc.com/2020/11/19/treasury-seeks-to-extend-some-emergency-fed-programs-but-end-others-including-main-street-facility.html">https://www.cnbc.com/2020/11/1...
In light of a recent GAO report that raises questions about recent rule changes involving PPP funds, Sec. Mnuchin and HHS CFO Moughalian should be asked: why transfer charges from CARES -> PPPHCE?
see:
#mt=e-report">https://www.gao.gov/products/B-332639 #mt=e-report
https://www.gao.gov/products/... href=" https://www.wsj.com/articles/ppp-was-a-fraudster-free-for-all-investigators-say-11604832072
https://www.wsj.com/articles/... href=" https://www.wsj.com/articles/sba-wins-temporary-delay-of-order-to-provide-details-on-ppp-borrowers-11605304678">https://www.wsj.com/articles/...
see:
#mt=e-report">https://www.gao.gov/products/B-332639 #mt=e-report
Taken together:
 https://abs.twimg.com/emoji/v2/... draggable="false" alt="🔸" title="Kleine orangene Raute" aria-label="Emoji: Kleine orangene Raute">Trump’s tweet is yet another example of him projecting bad deeds unto others
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🔸" title="Kleine orangene Raute" aria-label="Emoji: Kleine orangene Raute">Trump’s tweet is yet another example of him projecting bad deeds unto others
 https://abs.twimg.com/emoji/v2/... draggable="false" alt="🔸" title="Kleine orangene Raute" aria-label="Emoji: Kleine orangene Raute">Trump’s HHS *knowingly* purchased $175M+ worth of inaccurate tests, likely enabled by GSA
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🔸" title="Kleine orangene Raute" aria-label="Emoji: Kleine orangene Raute">Trump’s HHS *knowingly* purchased $175M+ worth of inaccurate tests, likely enabled by GSA
 https://abs.twimg.com/emoji/v2/... draggable="false" alt="🔸" title="Kleine orangene Raute" aria-label="Emoji: Kleine orangene Raute">Recent actions taken by senior officials at HHS and Treasury departments raise
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🔸" title="Kleine orangene Raute" aria-label="Emoji: Kleine orangene Raute">Recent actions taken by senior officials at HHS and Treasury departments raise  https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚩" title="Dreieckige Fahne an einem Pfosten" aria-label="Emoji: Dreieckige Fahne an einem Pfosten">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚩" title="Dreieckige Fahne an einem Pfosten" aria-label="Emoji: Dreieckige Fahne an einem Pfosten"> https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚩" title="Dreieckige Fahne an einem Pfosten" aria-label="Emoji: Dreieckige Fahne an einem Pfosten">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚩" title="Dreieckige Fahne an einem Pfosten" aria-label="Emoji: Dreieckige Fahne an einem Pfosten"> https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚩" title="Dreieckige Fahne an einem Pfosten" aria-label="Emoji: Dreieckige Fahne an einem Pfosten">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="🚩" title="Dreieckige Fahne an einem Pfosten" aria-label="Emoji: Dreieckige Fahne an einem Pfosten">
Fun fact— Alaska is one of the few states that is named specifically in the description field for the contracts related to this thread. Alaska received Cepheid tests (i.e. Abbott’s competitor).
(see https://projects.propublica.org/coronavirus-contracts/contracts/75A50120C00149)">https://projects.propublica.org/coronavir... https://twitter.com/kellyo/status/1330654857217404929">https://twitter.com/kellyo/st...
(see https://projects.propublica.org/coronavirus-contracts/contracts/75A50120C00149)">https://projects.propublica.org/coronavir... https://twitter.com/kellyo/status/1330654857217404929">https://twitter.com/kellyo/st...

 Read on Twitter
Read on Twitter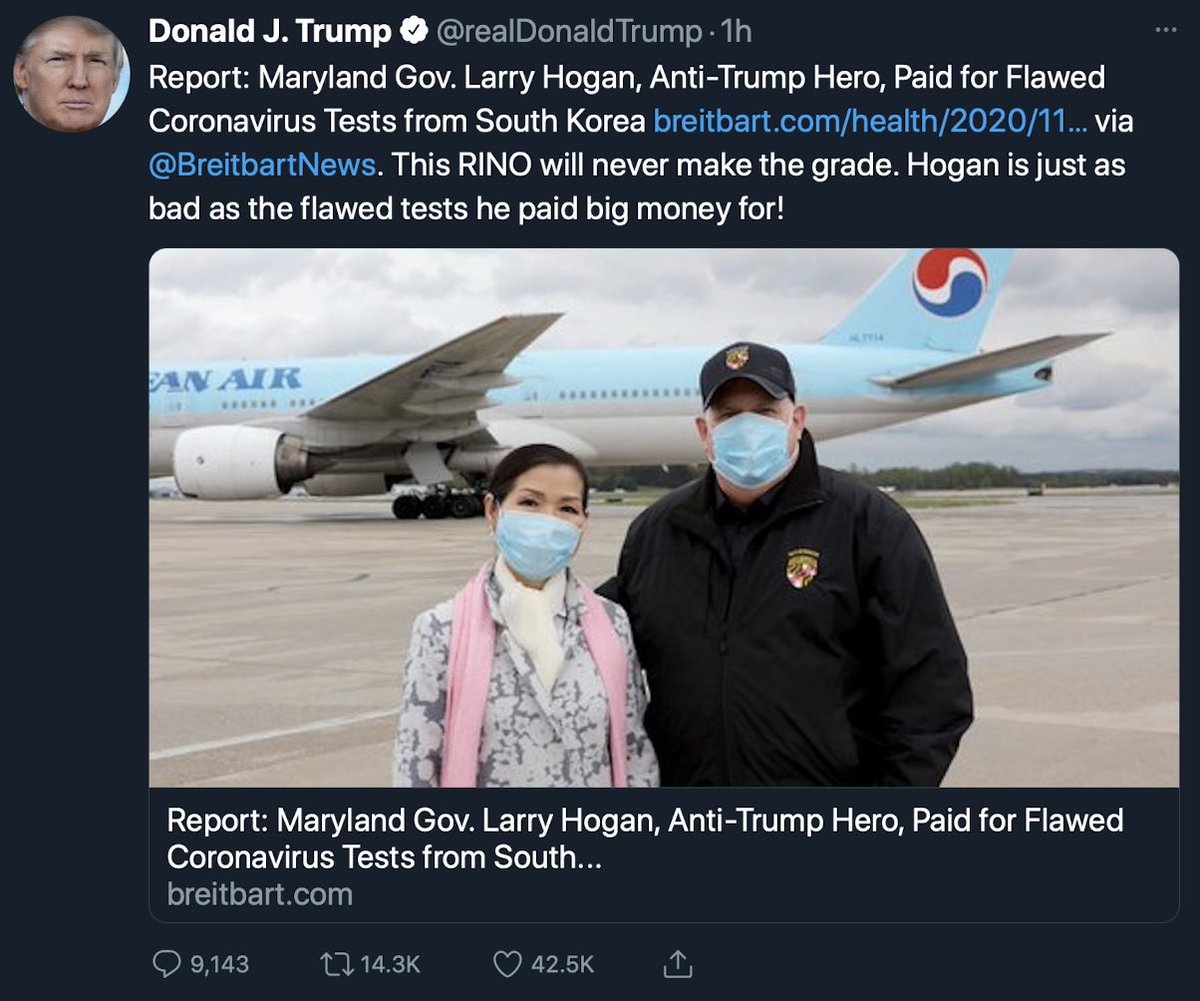 Investigative journalists would do well to dig into coordination between HHS, GSA, and Treasury on single-source/no-bid purchases of inaccurate #COVID19 tests totaling more than $175M+" title="https://abs.twimg.com/emoji/v2/... draggable="false" alt="👀" title="Augen" aria-label="Emoji: Augen"> Investigative journalists would do well to dig into coordination between HHS, GSA, and Treasury on single-source/no-bid purchases of inaccurate #COVID19 tests totaling more than $175M+" class="img-responsive" style="max-width:100%;"/>
Investigative journalists would do well to dig into coordination between HHS, GSA, and Treasury on single-source/no-bid purchases of inaccurate #COVID19 tests totaling more than $175M+" title="https://abs.twimg.com/emoji/v2/... draggable="false" alt="👀" title="Augen" aria-label="Emoji: Augen"> Investigative journalists would do well to dig into coordination between HHS, GSA, and Treasury on single-source/no-bid purchases of inaccurate #COVID19 tests totaling more than $175M+" class="img-responsive" style="max-width:100%;"/>
 https://projects.propublica.org/coronavir..." title="More to the point: Why was there such as disparity in the Trump Administration’s financial commitment to a faulty testing platform despite multiple public reports and an FDA notice?sources: https://projects.propublica.org/coronavir... href=" https://projects.propublica.org/coronavirus-contracts/vendors/abbott-rapid-dx-north-america-llc">https://projects.propublica.org/coronavir...">
https://projects.propublica.org/coronavir..." title="More to the point: Why was there such as disparity in the Trump Administration’s financial commitment to a faulty testing platform despite multiple public reports and an FDA notice?sources: https://projects.propublica.org/coronavir... href=" https://projects.propublica.org/coronavirus-contracts/vendors/abbott-rapid-dx-north-america-llc">https://projects.propublica.org/coronavir...">
 https://projects.propublica.org/coronavir..." title="More to the point: Why was there such as disparity in the Trump Administration’s financial commitment to a faulty testing platform despite multiple public reports and an FDA notice?sources: https://projects.propublica.org/coronavir... href=" https://projects.propublica.org/coronavirus-contracts/vendors/abbott-rapid-dx-north-america-llc">https://projects.propublica.org/coronavir...">
https://projects.propublica.org/coronavir..." title="More to the point: Why was there such as disparity in the Trump Administration’s financial commitment to a faulty testing platform despite multiple public reports and an FDA notice?sources: https://projects.propublica.org/coronavir... href=" https://projects.propublica.org/coronavirus-contracts/vendors/abbott-rapid-dx-north-america-llc">https://projects.propublica.org/coronavir...">






 https://www.acquisition.gov/far/part-..." title="While chatting with “along” and/or “awright” at HHS, it would be important to ask them *who* authorized the contract, and to confirm that this person had authority to do so. After all, that authority cannot be delegated under federal law. https://abs.twimg.com/emoji/v2/... draggable="false" alt="🤔" title="Denkendes Gesicht" aria-label="Emoji: Denkendes Gesicht"> https://www.acquisition.gov/far/part-..." class="img-responsive" style="max-width:100%;"/>
https://www.acquisition.gov/far/part-..." title="While chatting with “along” and/or “awright” at HHS, it would be important to ask them *who* authorized the contract, and to confirm that this person had authority to do so. After all, that authority cannot be delegated under federal law. https://abs.twimg.com/emoji/v2/... draggable="false" alt="🤔" title="Denkendes Gesicht" aria-label="Emoji: Denkendes Gesicht"> https://www.acquisition.gov/far/part-..." class="img-responsive" style="max-width:100%;"/>
 https://www.hhs.gov/about/age..." title="The only person who could legally authorize this contract is *Acting* Assistant Secretary for Financial Resources (ASFR) and *Acting* Chief Financial Officer for HHS, Jennifer Moughalian. see: https://www.hhs.gov/about/lea... href=" https://www.hhs.gov/about/agencies/asfr/key-personnel/index.html">https://www.hhs.gov/about/age...">
https://www.hhs.gov/about/age..." title="The only person who could legally authorize this contract is *Acting* Assistant Secretary for Financial Resources (ASFR) and *Acting* Chief Financial Officer for HHS, Jennifer Moughalian. see: https://www.hhs.gov/about/lea... href=" https://www.hhs.gov/about/agencies/asfr/key-personnel/index.html">https://www.hhs.gov/about/age...">
 https://www.hhs.gov/about/age..." title="The only person who could legally authorize this contract is *Acting* Assistant Secretary for Financial Resources (ASFR) and *Acting* Chief Financial Officer for HHS, Jennifer Moughalian. see: https://www.hhs.gov/about/lea... href=" https://www.hhs.gov/about/agencies/asfr/key-personnel/index.html">https://www.hhs.gov/about/age...">
https://www.hhs.gov/about/age..." title="The only person who could legally authorize this contract is *Acting* Assistant Secretary for Financial Resources (ASFR) and *Acting* Chief Financial Officer for HHS, Jennifer Moughalian. see: https://www.hhs.gov/about/lea... href=" https://www.hhs.gov/about/agencies/asfr/key-personnel/index.html">https://www.hhs.gov/about/age...">