Very interesting article:
"Applying lessons from the Ebola vaccine experience for SARS-CoV-2 and other epidemic pathogens"
 https://abs.twimg.com/emoji/v2/... draggable="false" alt="👇" title="Rückhand Zeigefinger nach unten" aria-label="Emoji: Rückhand Zeigefinger nach unten">
https://abs.twimg.com/emoji/v2/... draggable="false" alt="👇" title="Rückhand Zeigefinger nach unten" aria-label="Emoji: Rückhand Zeigefinger nach unten">
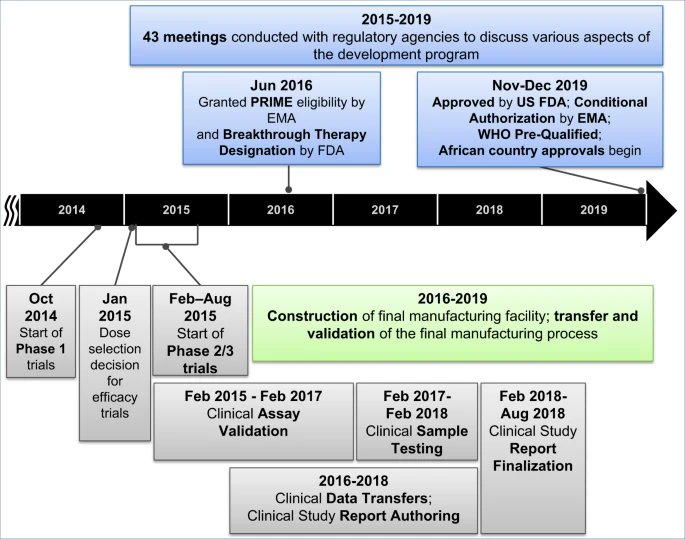
"The period of 5 years from the start of Phase 1 trials in Oct 2014 to the approval of this vaccine in Nov 2019... https://www.nature.com/articles/s41541-020-0204-7">https://www.nature.com/articles/...
"Applying lessons from the Ebola vaccine experience for SARS-CoV-2 and other epidemic pathogens"
"The period of 5 years from the start of Phase 1 trials in Oct 2014 to the approval of this vaccine in Nov 2019... https://www.nature.com/articles/s41541-020-0204-7">https://www.nature.com/articles/...
was much faster than the typical 10–15 year timeline for vaccine development and approval.
Extraordinary efforts were made to advance this vaccine candidate through Phase 1, 2, and 3 clinical trials and the data generated in the context of the West African Ebola outbreak"...
Extraordinary efforts were made to advance this vaccine candidate through Phase 1, 2, and 3 clinical trials and the data generated in the context of the West African Ebola outbreak"...
"has supported its licensure by the US Food and Drug Administration (FDA), conditional authorization by the European Medicines Agency (EMA) and several African countries, along with prequalification by the WHO".
"Through this Ebola vaccine development effort a number of learnings have been identified, which are highly relevant for the current vaccine development efforts in response to the COVID-19 pandemic."
Timeline of the development of the Ebola vaccine:
Timeline of the development of the Ebola vaccine:

 Read on Twitter
Read on Twitter