Our work on "Repurpose of a Chemosensory Macromolecular Machine" is out on @NatureComms today
https://www.nature.com/articles/s41467-020-15736-5
Here">https://www.nature.com/articles/... is a quick summary https://abs.twimg.com/emoji/v2/... draggable="false" alt="👇" title="Down pointing backhand index" aria-label="Emoji: Down pointing backhand index">(in English)
https://abs.twimg.com/emoji/v2/... draggable="false" alt="👇" title="Down pointing backhand index" aria-label="Emoji: Down pointing backhand index">(in English)
https://www.nature.com/articles/s41467-020-15736-5
Here">https://www.nature.com/articles/... is a quick summary
In Bacteria, chemosensory machines are a type of protein complex that helps bacteria respond to environmental cues with a variety of phenotypes.
Ref: https://www.nature.com/articles/nrm1524
The">https://www.nature.com/articles/... most famous chemosensory machine controls the flagellar motility in E. coli: the chemotaxis system.
Ref: https://www.nature.com/articles/nrm1524
The">https://www.nature.com/articles/... most famous chemosensory machine controls the flagellar motility in E. coli: the chemotaxis system.
In the cell, the proteins that compose these machines form macromolecular structures that we can see using #cryoEM
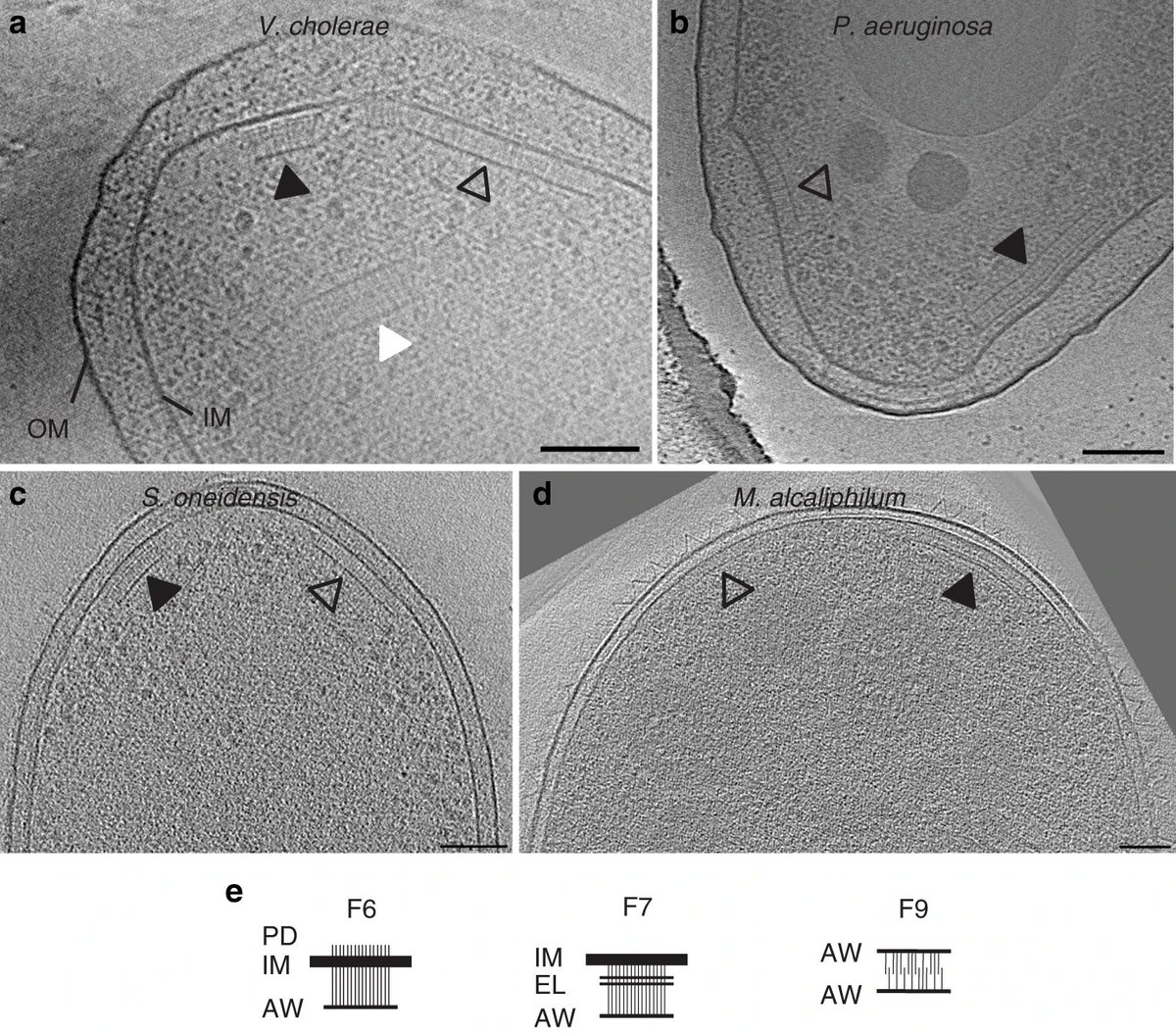
Imagining V. cholerae, P. aeruginosa, S. oneidensis and M. alcaliphilum under stress conditions, we noticed 2 or more chemosensory machines being expressed at once.
Imagining V. cholerae, P. aeruginosa, S. oneidensis and M. alcaliphilum under stress conditions, we noticed 2 or more chemosensory machines being expressed at once.
By doing mutagenesis in V. cholerae and P. aeruginosa, we determined that these arrays are formed by different chemosensory classes:
One is from the F7 class (the taller ones) and the other from the F6 class (the shorter ones).
One is from the F7 class (the taller ones) and the other from the F6 class (the shorter ones).
In these organisms, the F6 class controls flagellar motility. The function of the F7 class, in these bugs, remains unknown.
The F6 system has been studied before, so we took a closer look at the ultrastructure of the F7 system.
The F6 system has been studied before, so we took a closer look at the ultrastructure of the F7 system.
Only to find that the domain architecture of the chemoreceptors that form the F7 array makes electron densities that can be resolved by #cryoEM.
Even the famous PAS domains.
Even the famous PAS domains.
The presence of F7 and F6 systems in all these organisms suggests that the last common ancestor of Gammaproteobacteria had both systems. And the F6 controls flagellar motility.
But what about in E. coli? Which system controls flagellar motility?
But what about in E. coli? Which system controls flagellar motility?
Surprisingly, the flagellar motility is controlled by the F7 system in E. coli.
E. coli does not even have F6 systems.
How did the F7 system learn to control flagellar motility in Enteric bacteria like E. coli and Salmonella?
E. coli does not even have F6 systems.
How did the F7 system learn to control flagellar motility in Enteric bacteria like E. coli and Salmonella?
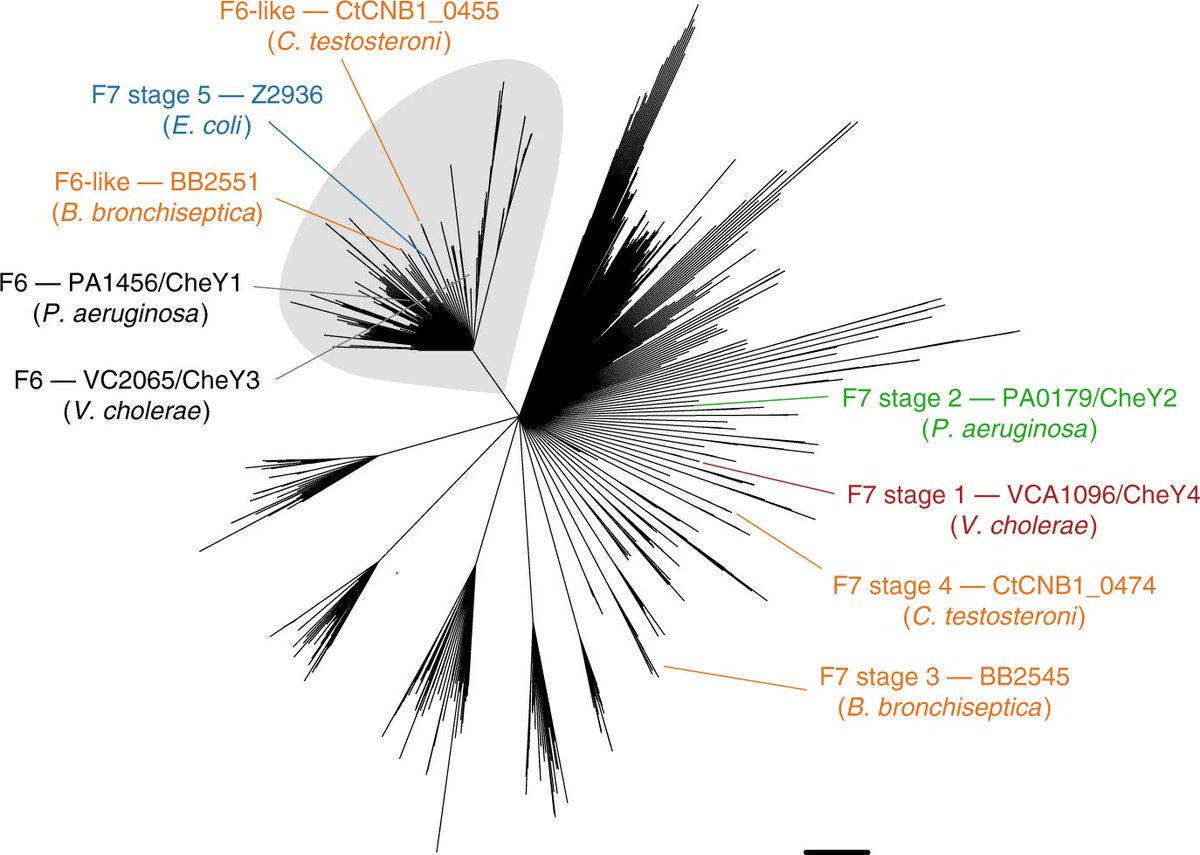
We used comparative genomics to map stepwise changes in the F7 gene cluster in Gamma and Betaproteobacteria.
We pinpointed major evolutionary changes in the cluster organization that ultimately lead to the repurposing of the F7 system to control flagellar motility.
We pinpointed major evolutionary changes in the cluster organization that ultimately lead to the repurposing of the F7 system to control flagellar motility.
So what happened?
It appears that the F6 system was lost, and the F7 system sequestered the output protein from the F6 system: a response regulator (CheY) that binds to the flagellar motor switching rotational bias.
It appears that the F6 system was lost, and the F7 system sequestered the output protein from the F6 system: a response regulator (CheY) that binds to the flagellar motor switching rotational bias.
By doing comparative analysis, we showed that CheY adapted to the F7 system by changing residues in the interface of interaction with the histidine kinase (CheA) of the F7 system.
The other important change was the input of the system (the chemoreceptor).
It switched from those multi- cytoplasmic domains we mentioned before, to a simpler topology: a periplasmic input domain, a HAMP, and a signaling domain. (just like the receptors in the F6 system).
It switched from those multi- cytoplasmic domains we mentioned before, to a simpler topology: a periplasmic input domain, a HAMP, and a signaling domain. (just like the receptors in the F6 system).
So, what did we learn?
We learned that the F7 system of unknown function in ancestral Gammaproteobacteria was repurposed to control flagellar motility in Enteric by 2 major changes:
Input and output.
We learned that the F7 system of unknown function in ancestral Gammaproteobacteria was repurposed to control flagellar motility in Enteric by 2 major changes:
Input and output.
If we think that the chemosensory system in bacteria is an analog of control systems, it makes sense that it could be repurposed by changing what it senses (input), what it acts on (output), and minor tweaks in the dynamics of the system.
Our results suggest that chemosensory might be a good candidate to modulate cellular functions that require sophisticated control in bioengineered bacterial "robots".
They come out of the box with serious features: adaptation, memory, signal integration, and more.
They come out of the box with serious features: adaptation, memory, signal integration, and more.
Many thanks to everyone involved in this work: @theJensenLab, @BriegelAriane, @RinggaardSimon, @koogy27, and other incredible twitterless authors.
Also, thanks to the reviewers that truly pushed us to a better paper. Including anons, G. Hazelbauer, and @KrellLaboratory.
Cheers!
Also, thanks to the reviewers that truly pushed us to a better paper. Including anons, G. Hazelbauer, and @KrellLaboratory.
Cheers!

 Read on Twitter
Read on Twitter